Find out how LabCollector can help you as a COVID test lab!
The year 2020 was marked by an unprecedented event. A pandemic caused by the coronavirus SARS-CoV-2, more commonly known as COVID-19 (for CoronaVIrus Disease) struck the world with daunting effects. Since the beginning of the year the number of new cases of COVID-19 were and are still increasing. As reported by the WHO, the total number of people affected is now more than 64.3 million and 1.4 million deaths. (result as of December 04, 2020)
Widespread COVID researchers, testing labs, public health organizations, private sector businesses joining to help combat to control the coronavirus pandemic sweeping across the world. More than 15 million PCR tests have been sent to laboratories around the world (WHO, Weekly Operational Update on COVID-19, November 30, 2020). On daily bases, these labs and testing centers need to EFFICIENTLY manage large scale number of tests arriving in addition to their other common and routine tests. They must also ensure FAST delivery of test results to patients to limit the spread of the virus. However, handling of such large number of tests daily is another hurdle. LIMS (Laboratory Information Management Systems) have proven to be a boon in such difficult period.
Our full featured LabCollector LSM Pack qPCR Automation has helped COVID testing labs manage the unprecedented acceleration of laboratory workflow from test requestions to sending reports, ensure compliance, including HIPAA and other privacy concerns.

LabCollector LSM pack deliver as a comprehensive Software-As-A-Service (SaaS) solution, hosted in the cloud requires no infrastructure investment. We do the configuration and set up to make it a turnkey with all the attribution of your workflow requirements with minimal implementation time to go-live in production as quick as possible in less than four weeks.
Moreover, LabCollector offers an effortless and complete laboratory management experience giving you the ability to enter biospecimens into a biobank and rapidly begin conducting COVID-19-related testing and research. Also, expand your LIMS capability with many extensive Add-Ons such as Electronic Laboratory Notebook, WorkFlow Manager, Equipment Scheduler, Gantty project, and many more as well as consumables management, labeling and barcoding. The package also includes, training to get up and running quickly with ongoing support services.
They trusted us to help them in the fight against COVID-19

LABCOLLECTOR CAN HELP YOU AT EVERY STEP!
1. Requesting your test made easier with LSMRemote web portal
Laboratories performing COVID tests receive many test requests from different collection centers every day. In order to facilitate the whole process from requisition to result reporting, we have developed a free ready-to-use web portal, LSMRemote.
Through this portal requesters/patients can easily register and even pre-register details of test request, patients’ demographics, sample information, collection date/time…etc. Tests labs using LabCollector software on the Lab Service Management add-on (LSM) receive the job request and can manage the jobs, perform the test, send custom reports to requesters and/or to the patients. LSMRemote also provides access portal to patients to easily view the status of the test and download their report as soon as it is available.
Thus, for the COVID testing lab this process instructions to be carried out on a single tool where everything is centralized increases efficiency of timeline that were unworkable given the rapidly escalating pandemic and its alarming infection rates.
“DIRECT CONNECTION WITH YOUR REQUESTERS FOR TESTS REQUESTS”
 |
At your collection sites, to make your sample collection and associated sample information streamlined and error proved, we have developed pre-printed labels to  The test/patient information is then linked to this unique barcode throughout the test chain. |
Requesters can have assurance of their test request and sample received by obtaining the Chain of Custody (CoC) report. This is an additional proof of quality regarding the chain of custody and transport of samples.
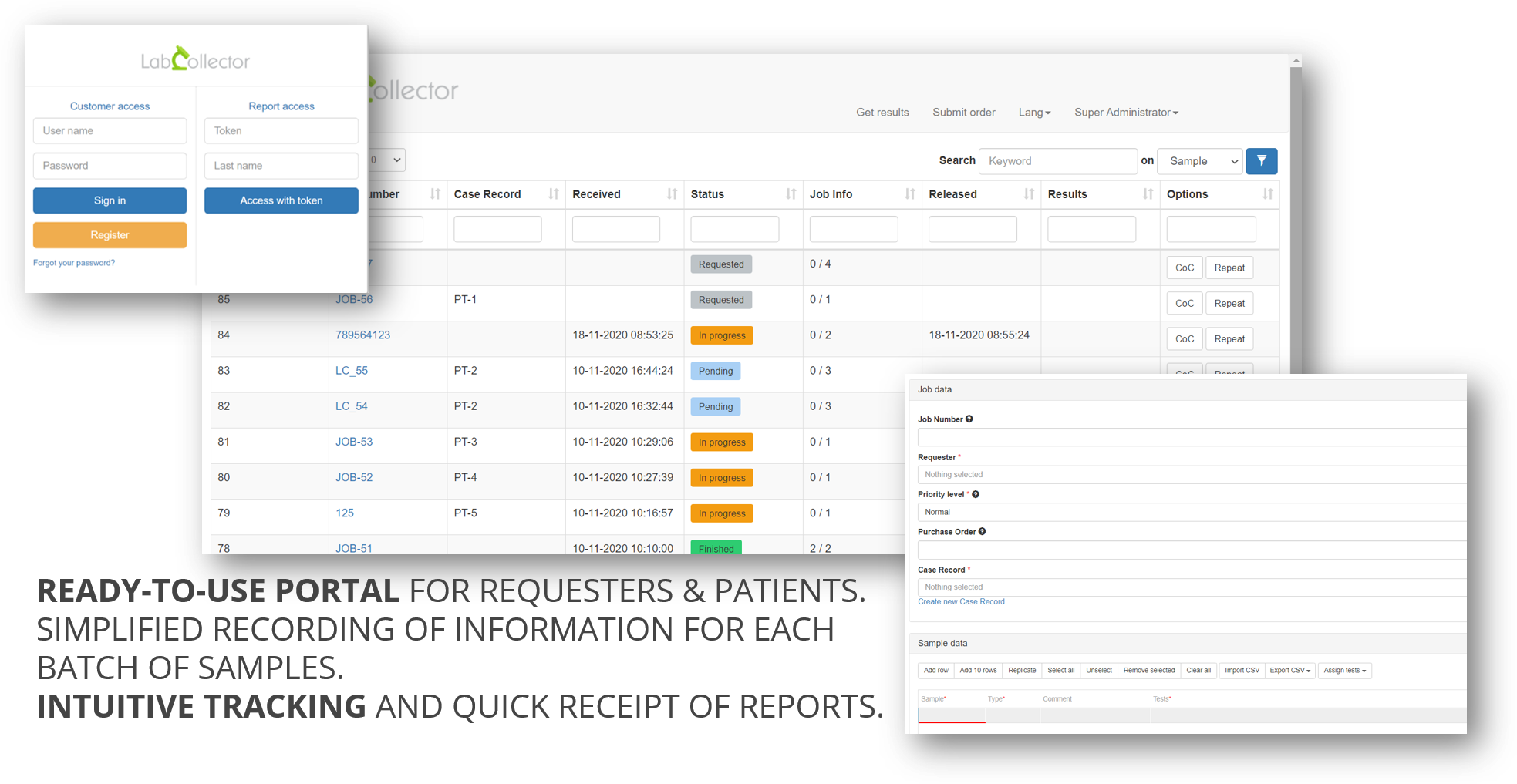

A new option is now available on LSMRemote portal. LSMRemote now provides option for patients to pre-register before providing their samples. The information about the patient can be pre-registered by the patient. These fields in the Patient pre-registration form can be configured in custom made Patient module. Once the pre-registration by patient is done on the form, this information is automatically integrated into the custom Patient module in LabCollector which can be then connected to the LSM add-on with the help of case record function. Once the patients have registered, they can provide their samples in the clinic, drive-thru or sample collection center.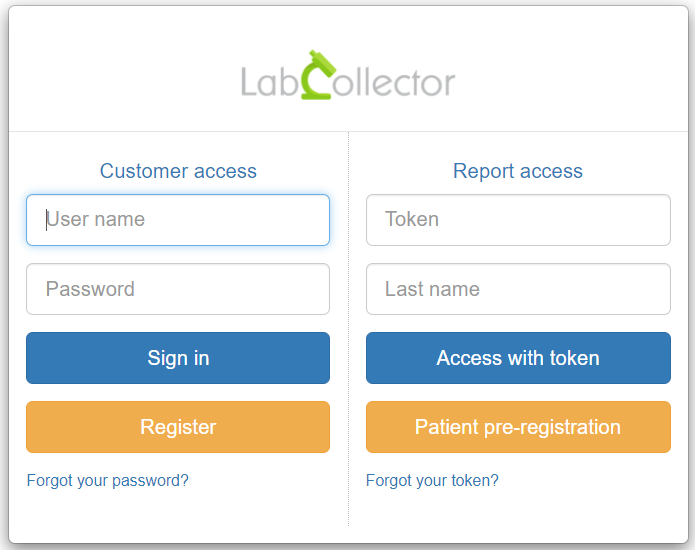

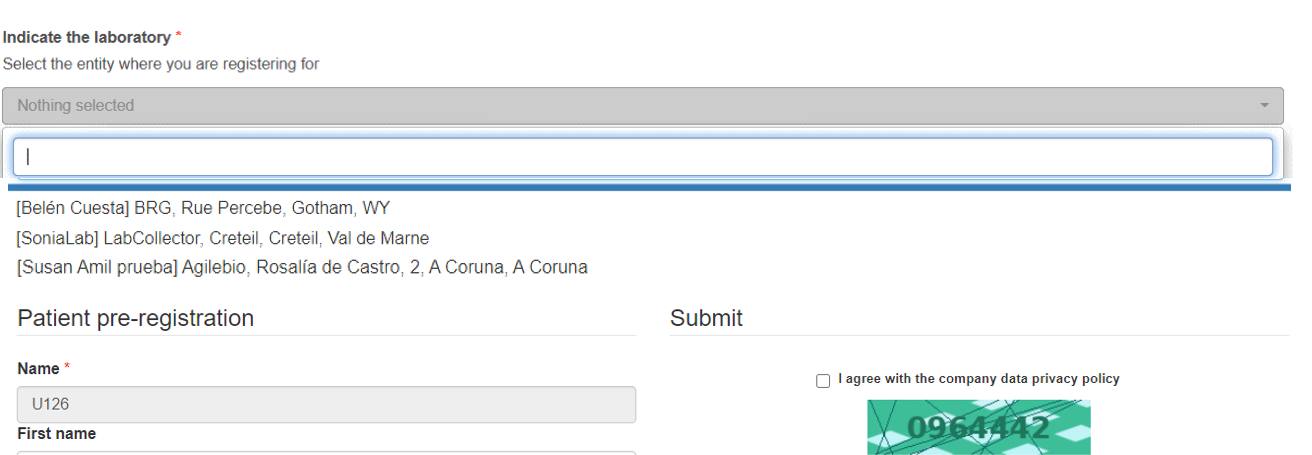
In order to efficiently track the origin of samples and thus increase compliancy, it is now possible during patient pre-registration to select the collection lab (requester/provider) in the new section “Indicate the laboratory” (see picture below). This option allows the patients to choose the requester/provider and at the same time the requesters/providers will not be able to see patients (case records) of other requesters/provider.

2. Speed up your samples receiving process
When samples arrive in test labs, it is usually individual single sample or in batches of tens or even hundreds, or thousands of samples at the same time. To improve the speed of reception and avoid manual input errors, LabCollector provides several tools to receive samples quickly and easily.
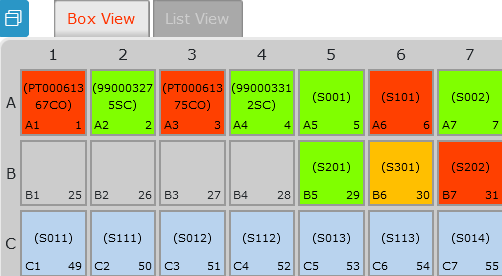
- Samples module: This basic module allows you to receive samples by unit or by batch. You store any kind of samples and create a link to LSM and other add-ons.
- Sample receiving add-on: With this add-on you can accelerate sample accession and unpacking a large quantity of packages of samples received. It gives you the ability to perform quality analysis and checks on samples to verify samples before adding to plates and creating your sample plate map. Within the Sample Receiving add-on, you can manage the storage of plates/boxes and you can also start templates in the Workflow Manager add-on. The Sample Receiving add-on works with the Lab Services Manager add-on (LSM) and an external API in order to check sample barcodes or status. The Sample Receiving and LSM add-on supports the barcode system so that you can print barcodes or read barcodes already affixed to incoming packages and samples.
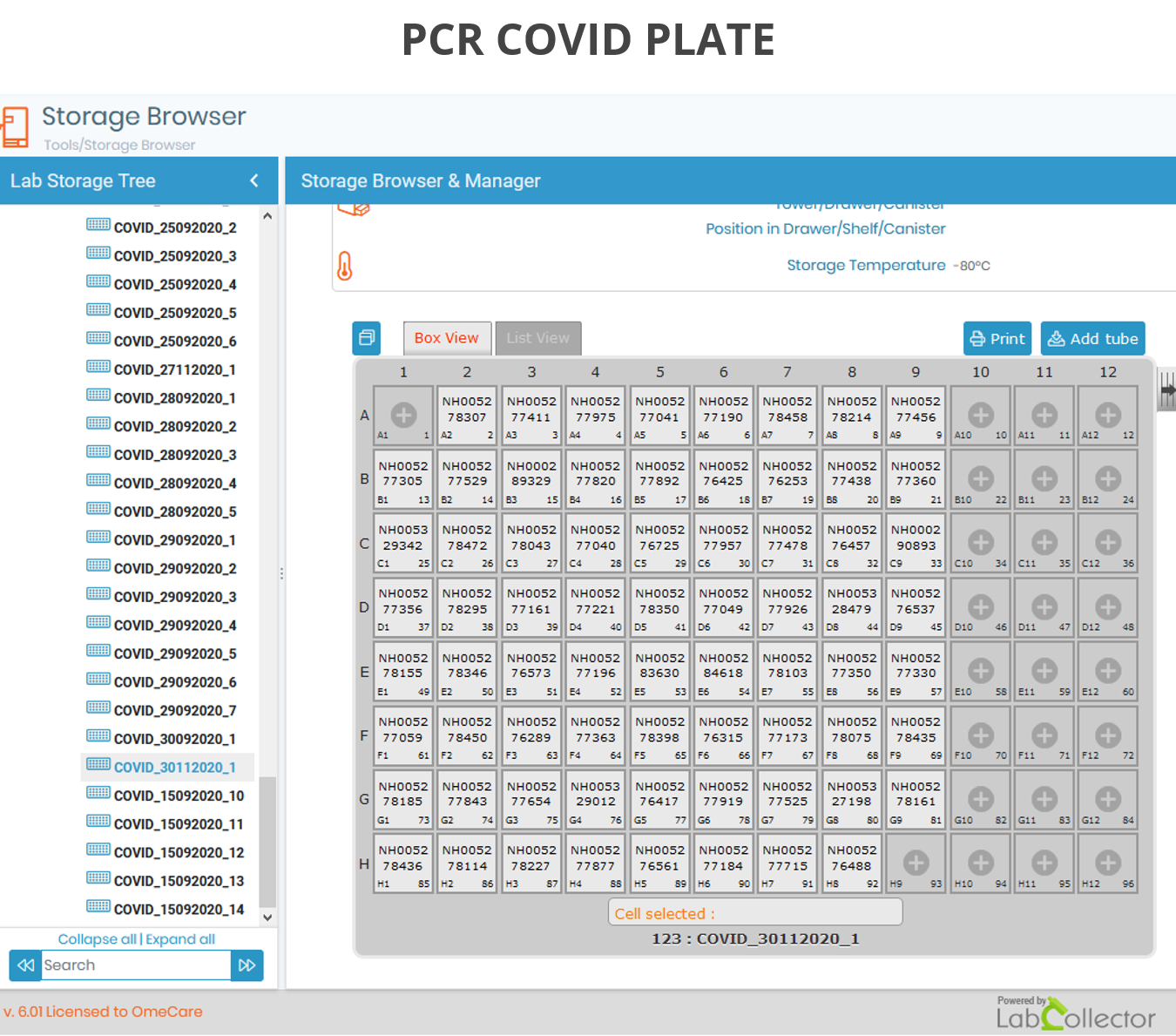
All of this allows you to create and manage a hierarchical and organized storage, ideal for retrieving all your samples anywhere, anytime as you can see on the picture below. It’s a considerable time saver!

   |
Thanks to the LabCollector Data Logger add-on, you can also monitor all the parameters of your instruments (temperature, gas, light, humidity, etc.) and be alerted in case of damage. Thus, the quality of your samples is never endangered and a quality follow-up can be carried out thanks to regular data logging. This monitoring of environmental parameters is crucial as an analytical laboratory is always at the fragility of damaging temperature sensitive samples and the importance of conservation for the reliability of the tests. |
3. I-Collector for automatic instrument integration
Once all samples have been received, the requested tests must be carried out. To do so, we have I-Collector tool to automate tests by sending commands directly from LabCollector. This tool acts as middleware and allows a bi-directional communication between LabCollector and your instruments. I-Collector supports a growing number of instruments but we offer the development of any plugin needed for any of your lab instrument equipment. Results and values are automatically sent back to LabCollector or the LSM add-on as soon as the instrument releases them.
“YOUR EQUIPMENT INTEGRATED DIRECTLY TO LABCOLLECTOR
THANKS TO I-COLLECTOR”


I-Collector benefits from our API/Web-Service engine. The LabCollector API allows you to read and write data from/to any instrument in your lab, thus guaranteeing data integrity and minimizing execution time.


This integration allows considerable time savings on the realization of large-scale tests. Laboratories that have to follow FDA or ISO regulations must be more vigilant in their workflow, so automating the collection of results helps to avoid errors during multiple file manipulations. Learn more about I-Collector on our blog.
   |
With the LabCollector Worflow Manager add-on, you can orchestrate your data and lab servicing with a graphical flow chart designer. This add-on allows the management of workflow templates (a catalog of job workflows) to start new jobs. All jobs are automatically stored which allow for monitoring status and advancement. This add-on can be configured to run production batches, clinical trials procedures and any lab operation like COVID test.
|
4. Analysis of results and reporting is more efficient
When a job is completed, you can generate a result report that has been previously validated by a “validator”. This validation step may be necessary to ensure that the entire process complies with established standards.
Our expert team of developers set up programming logics based on customer-determined instrument output result files, report templates for testing using a variety of protocols, such as RT-PCR and serology, or any others. At the end of the test, the results are sent automatically to LSM and instrument reports are generated so that they can be sent automatically to patients or local/state authorities (see below-part 6).
In case of errors or tests to be repeated for a patient, an option available on LSMRemote allows the requester to repeat a test with the same samples or new samples that will then be sent to the analysis laboratory. This request is then transmitted directly to the analysis lab, which can then act on the request.


In LabCollector, you can fully configure the report template by displaying the data you want such as the case record, patients’ demographics, the test, the result, the comment, signature, etc.
From a more administrative point of view, the costs of a test can be configured to automatically create a quote or invoice for each request. At the end of a job, an invoice is created with the tests performed as well as the cost of each test previously configured. The invoice template is also configurable.
This invoice (or quote) can be sent directly via LSMRemote to the requesters, allowing easy administrative management of the laboratories.
Thus LSMRemote portal saves time and considerably increases efficiency in the management of test requests but also in the administrative management of documents between the parties.
   |
New tags are now available for report. This new feature allows you to amend or correct a finished/validated reports. You can also add append comments for LSM parameters now with our new tags. Specially, when a report result is invalid/inconclusive you can add the comment (reason) for the same in you report.
Visit our Knowledge Base page to learn more about this new feature. |
5. Provision of results thanks to LSMRemote
Through the LSMRemote portal patients/requesters who has requested COVID-19 test are able to see the status/progress of the test, the completed results, and validated reports.
On this portal, patient can simply login with a unique identifier (token). This identifier is created at the time of sample collection and recording of information on LSMRemote and is directly transmitted to the patient for later use. Thus, the connection and the transmission of results is secure and expedited for COVID-19 diagnosis.
6. Reporting to Local/State Authorities
At this time of pandemic, an expedited ramp-up in COVID-19 diagnostic testing is immediate in order to take adequate measures, especially now that the number of COVID cases seems to be increasing. Therefore, test reports based on standard administrative, CDC reporting requirements such as the number of positive or negative results and the total number of people tested, etc. must be sent to the Local and State authorities.
“DIRECT CONNECTION AND REPORT SENDING TO THE AUTHORITIES”
In order to facilitate this administrative step and to have a rigorous follow-up of the data, we have developed a special plugin for our I-Collector solution allowing to execute the sending of a report at a predefined and precise regular interval. It is thus possible to generate an HL7 message (Health Level Seven : set of international standards for transfer of clinical and administrative data) with a specific data mapping and to send it automatically by the SFTP or SOAP protocol (secured data transmissions) to the administrations.
This tool is currently used in the United States, but it is possible to adapt it to a completely different situation and thus save a considerable amount of time in the execution of this task.
How our LabCollector LIMS can help in COVID-19 crisis?
Well, on several points…initially LabCollector is there to help laboratories in different fields in the management of their laboratory. Indeed, in these dire times we also join our hands and fight together against COVID-19 and made LabCollector the available tool by allowing to increase the integration of your lab and the centralization of information on a single tool solution.
“WITH LABCOLLECTOR YOUR WORK IS MORE EFFICIENT AND PRODUCTIVE”
Your daily life will be easier to manage and your work will be more efficient and productive. Don’t panic anymore when hundreds of samples to be tested arrive, the transcription of results from your equipment, the timely sending of reports for patients, the sending of invoices and reports to administrations. Everything is under control! No more painful process! LabCollector and the entire AgileBio team is here to ensure rapid deployment and support you throughout this change.
Thanks to our LIMS, you can now offload some tasks and automate some of them in order to focus on what is really important: your analysis work!


To help set up LabCollector v6.01 please refer to our Manuals and our Knowledgebase.
Image Credits: Pixabay
Graphics Credits: Freepik, Font Awesome